Quality Management System
Quality Management
End-to-End Quality Control
Our commitment to quality assurance, regulatory compliance, and advanced manufacturing systems positions us as a trusted partner for companies seeking scalable, custom-developed cosmetic solutions—not private label or relabeling, but true formulation and product innovation from the ground up.
All Global Cosmetics manufacturing facilities are certified by CNAS (China National Accreditation Service for Conformity Assessment) and operate in compliance with international cosmetic manufacturing standards. This ensures that every product developed through our OEM/ODM process meets strict global requirements for:
- Product safety
- Ingredient traceability
- Stability and compatibility
- Microbiological control
- Regulatory readiness for international markets
Our in-house laboratories and formulation teams are equipped with cutting-edge testing technologies and supported by highly trained engineers and technicians. We implement precision-driven quality management systems throughout every stage of development and production, ensuring:
- Consistent formulation performance
- Scalable manufacturing accuracy
- On-time production delivery
- Full batch traceability
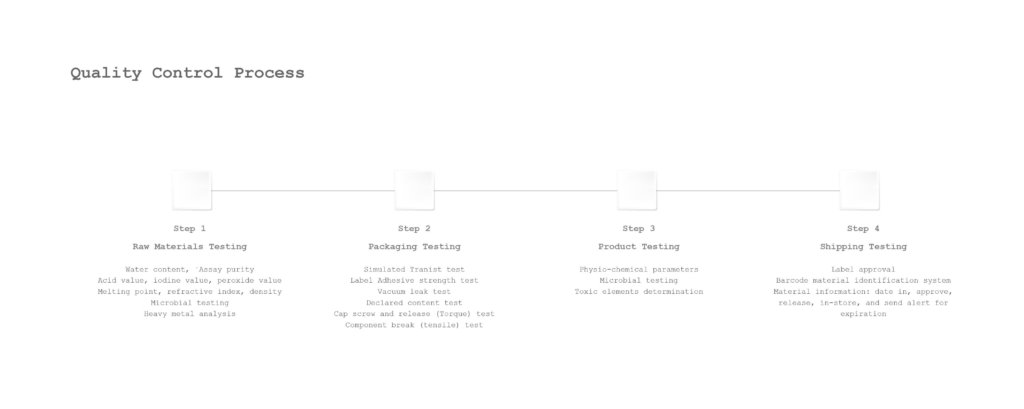
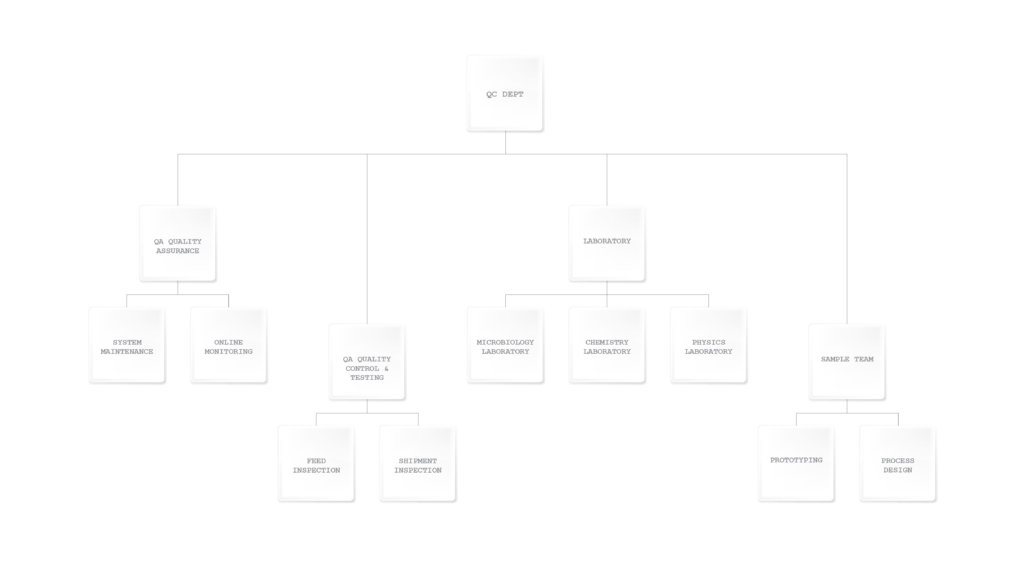
Our Core Quality Control Systems Include:
1. Full Barcode Traceability System
Every raw material and finished product is tracked through an end-to-end barcode system, ensuring complete visibility across sourcing, production, and distribution.
2. Continuous In-House Quality Assurance
Our engineers conduct real-time quality monitoring across all production lines and warehouse operations to maintain strict consistency standards.
3. Strict Hygiene & GMP Protocols
We follow documented hygiene, sanitation, and maintenance procedures aligned with global GMP expectations to ensure contamination-free production environments.
4. Rigorous Product Inspection Standards
All products undergo thorough inspection protocols, including:
- AQL Level II sampling
- 100% inspection where required
- Zero tolerance for microbial contamination or functional defects
Audit Certifications





We are here for all your business needs and questions.
Please feel free to contact us on your queries and we will get back within 24 hours.